Reverse Osmosis
Reverse Osmosis: Imitating a natural phenomenon
Salts have the tendency to absorb water. In nature the feeding of organisms occurs via the transport of mineral salts and acids dissolved in water passing through the cell membrane thanks to osmotic pressure, until the cellular pressure and osmotic pressure equalize.
In Reverse Osmosis, water with salts dissolved at a higher pressure than osmotic pressure passes through a membrane permeable only to pure water, resulting in a current of desalinated water (permeate) which is collected, while the remaining solution with a high saline content (concentrate) is expelled.
How it works
 To explain the principle of Reverse Osmosis, one must first understand the phenomenon of osmosis. Osmosis can be defined as "a process by which molecules of a solvent tend to pass through a semi-permeable membrane (such as those of living cells) from a less concentrated solution into a more concentrated one, thus equalizing the concentrations on each side of the membrane." (See Fig. 1)
To explain the principle of Reverse Osmosis, one must first understand the phenomenon of osmosis. Osmosis can be defined as "a process by which molecules of a solvent tend to pass through a semi-permeable membrane (such as those of living cells) from a less concentrated solution into a more concentrated one, thus equalizing the concentrations on each side of the membrane." (See Fig. 1)
 The vessel on the left is filled with water, and the tube is half-submerged in the water.The level of water in the tube is the same as the level of water in the vessel. In the central diagram the lower end of the tube has been closed off with a semi-permeable membrane and the tube has been partially filled with a saline solution and immersed. Initially the levels of the saline solution and that of the water are equal, but after a while the level of liquid inside the tube rises.This increase is due to "osmotic pressure". A semi-permeable membrane is a membrane that allows certain atoms or molecules to pass while blocking others. In Fig. 2, the membrane allows the water molecules to pass but not the salt molecules.
The vessel on the left is filled with water, and the tube is half-submerged in the water.The level of water in the tube is the same as the level of water in the vessel. In the central diagram the lower end of the tube has been closed off with a semi-permeable membrane and the tube has been partially filled with a saline solution and immersed. Initially the levels of the saline solution and that of the water are equal, but after a while the level of liquid inside the tube rises.This increase is due to "osmotic pressure". A semi-permeable membrane is a membrane that allows certain atoms or molecules to pass while blocking others. In Fig. 2, the membrane allows the water molecules to pass but not the salt molecules.
A model to understand osmotic pressure could be one to think that the water molecules are to be found on both sides of the membrane.The molecules are in continuous movement (Brownian motion). On the side with the salt water, some of the openings are obstructed by the salt molecules while on the other side this does not happen.
 In this situation an higher number of water molecules passes from the water side to the salt side because there are more openings free for the passage. The level on the salt side increases until one of the following situations occurs: the saline concentration becomes equal on both sides of the membrane (impossible if on one side there is pure water and saline solution on the other); the pressure of the solution increases, in proportion to the height increase of the salt solution column, until it is the same as osmotic pressure.
In this situation an higher number of water molecules passes from the water side to the salt side because there are more openings free for the passage. The level on the salt side increases until one of the following situations occurs: the saline concentration becomes equal on both sides of the membrane (impossible if on one side there is pure water and saline solution on the other); the pressure of the solution increases, in proportion to the height increase of the salt solution column, until it is the same as osmotic pressure.
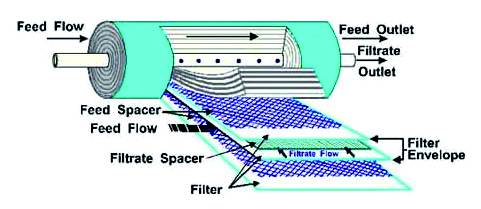
At this point the osmosis gives no further visible effects. In Reverse Osmosis, applying a pressure to the more concentrated solution, the natural flow direction between two saline solutions separated by a semi-permeable membrane is inverted. In practice, the semi-permeable membranes, which are made of synthetic materials and installed in distinct groups (see Fig. 3), are the means by which this process is rendered possible.The water to be treated enters the separation module and exerts sufficient pressure on the membranes.
The part of the water deprived of salts (permeate), passes through the membranes and is collected in the permeate storage area.The remaining, more concentrated, part of the water leaves the module having passed through it completely. A minimum continuous flow of concentrate is fundamental to guarantee the flushing of possible deposits and the absence of salt precipitation.
Salts have the tendency to absorb water. In nature the feeding of organisms occurs via the transport of mineral salts and acids dissolved in water passing through the cell membrane thanks to osmotic pressure, until the cellular pressure and osmotic pressure equalize. [See Fig. 3]
Function and equipment
The Reverse Osmosis systems are equipped with the following safety features:· Protection against dry working:
If the first quantity of input water in arrival does not reach the minimum pressure the apparatus shuts off automatically
· Manometers:
Useful to check the pump performance or blockage of the membrane
· Flow gauge:
Useful to check the flow of the permeate and the concentrate
· Pressure gauge:
Useful to check the input pressure of the water to be treated
· Conductivity control?
The conductivity is constantly measured by a conductivity cell and the relative value is sent to the central programmer controlling the rates relative to pre-established maximum and minimum set points. Maintenance of osmosis systems is extremely simple, all the components are easily accessible, so that cleaning, checking and parts exchange can take place without any problems.
Pre-treatment
Normally this process takes place without the use of chemical products. Thus Reverse Osmosis may be considered a highly ecological and econo- mical process.A preliminary pre-treatment of the water is only necessary if iron, manganese, chlorine or oily substances are present, as these substances may block the membrane.While our Reverse Osmosis systems are usually equipped with an initial micro-filtration process and a receptacle for antiscalant products, occasionally it is necessary to insert an activated carbon de-chlorination filter or a resin exchanging column for iron and manganese. Each of these systems noticeably extends the life of the membrane.Possible uses
Thanks to its numerous and indisputable qualities, Reverse Osmosis is already used in many sectors, such as drinking water production, the preparation of water for boilers, for refrigeration and for industrial processes. Many machines are in service in municipal drinking water networks, in hotels, hospitals, the cosmetics and pharmaceutical industries, banks and insurance.Working Conditions
Thanks to its numerous and indisputable qualities, Reverse Osmosis is already used in many sectors, such as drinking water production, the preparation of water for boilers, for refrigeration and for industrial processes. Many machines are in service in municipal drinking water networks, in hotels, hospitals, the cosmetics and pharmaceutical industries, banks and insurance.Working Conditions
Supply water at 15 °CMax. salinity 1000 ppm
SDI < 3
Permeate counterpressure 0 bar
Entry pressure 2-6 bar
Yield 50 - 75%
Salt retention min. 95%



